Abstract
Introduction Myelodysplastic syndromes (MDS) are heterogeneous hematopoietic neoplasms characterized by peripheral blood cytopenia and increased risk of evolution into acute myeloid leukemia. Recently, a new clinical-molecular prognostic model (International Prognostic Scoring System-Molecular, IPSS-M) was proposed to improve conventional risk stratification defined by the IPSS-R [Bernard E et al, NEJM Evid 2022]. In this study, we evaluated the clinical implementability of IPSS-M by: i) providing an extensive independent validation in a real-world patient population; ii) investigating the predictive and prognostic power of IPSS-M in patients receiving disease-modifying treatments (hypomethylating agents, HMA and allogeneic stem cell transplantation, HSCT), and iii) testing the accuracy of IPSS-M prediction when molecular information is missing to define a minimum set of relevant genes associated with high performance of the score.
Patients and Methods Clinical, cytogenetic and molecular data were collected at diagnosis and/or before starting disease modifying treatment in 2,876 MDS patients from GenoMed4All consortium (Genomics and Personalized Medicine for all through Artificial Intelligence in Haematological Diseases - www.genomed4all.eu). Survival curves were estimated with the Kaplan-Meier method. Multivariate analyses were performed by Cox's proportional hazards regression models. Model discrimination was assessed using Harrell's concordance index (C-index).
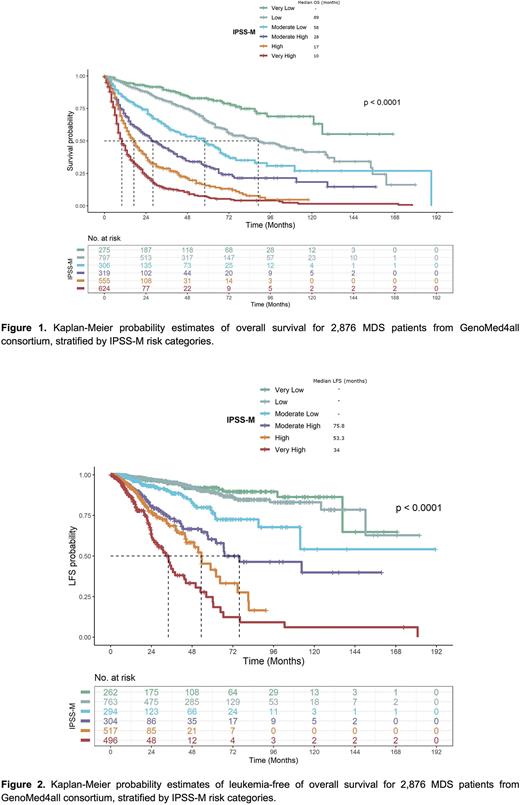
Results We calculated the IPSS-M score in the study cohort according to clinical and molecular features collected at diagnosis. Accordingly, 9.6% of patients (n=275) were classified as Very-Low risk, 27.7% (n=797) as Low risk, 10.6% (n=306) as Moderate-Low risk, 11.1% (n=319) as Moderate-High, 19.3% (n=555) as High risk and 21.7% (n=624) as Very-High risk, resulting in the re-stratification of 46% of patients with respect to IPSS-R. Compared to the IPSS-R, the IPSS-M led to an improved discrimination across all endpoints (C-index was 0.81 vs. 0.74 for overall survival, OS and 0.89 vs. 0.76 for leukemia-free survival, LFS, respectively, Figure 1 and2).
We then analyzed the predictive/prognostic value of IPSS-M in patients receiving HSCT (n=964) and HMA (n=268), respectively, in which clinical and molecular data were available before starting treatment. In patients treated with HSCT, IPSS-M significantly improved the prediction of the risk of disease relapse and the probability of post-transplantation survival with respect to IPSS-R (C-index was 0.76 vs. 0.60 for OS and 0.89 vs. 0.70 for probability of relapse, respectively). The prognostic effect of the IPSS-M was maintained in a multivariate analysis including recipient age and sex, time from diagnosis to transplantation, source of hematopoietic stem cells, type of donor, disease status at transplant and conditioning regimen as covariates (HR was 1.18 for OS and 1.38 for probability of relapse, P<.001). In patients treated with HMA, the probability of overall response evaluated according to 2006 IWG Criteria was 42%, without significant difference among IPSS-M categories (P= 0.19). Median OS in the whole population treated by HMA was 13.9 months. The median probability of OS was 20.7 months in Moderate-High, 17.9 months in High, and 12.7 months in Very-High risk patients (P<.001).
Finally, we analyzed the loss of accuracy of IPSS-M prediction in the study population when one or more IPSS-M-related molecular features were missing. This analysis is expected to provide useful information for clinicians with respect to the robustness of personalized prognostication when providing IPSS-M calculation with missing values. Information on mutational status of a set of 15 genes (ASXL1, CBL, DNMT3A, ETV6, EZH2, FLT3, IDH2, MLLPTD, NPM1, NRAS, RUNX1, SF3B1, SRSF2, TP53multihit and U2AF1) was required to obtain an accuracy of IPSS-M prediction of 80% (with a C-index of 0.8), while considering a set of 10 and 7 genes the accuracy of IPSS-M prediction decreased to <70% and <60%, respectively.
Conclusion IPSS-M improves MDS prognostication and might result in a more effective selection of candidates to HSCT. Additional factors other than gene mutations can be involved in determining HMA sensitivity. The definition of a minimum set of relevant genes may facilitate the clinical implementation of the score.
Disclosures
Meggendorfer:MLL Munich Leukemia Laboratory: Current Employment. Giaccone:MSD: Honoraria; MSD: Honoraria; Gilead: Honoraria; Novartis: Honoraria; MSD: Honoraria. Voso:Astellas: Speakers Bureau; Celgene/BMS: Consultancy, Honoraria, Research Funding, Speakers Bureau; Novartis: Research Funding, Speakers Bureau; jazz: Consultancy, Speakers Bureau. Oliva:Novartis: Patents & Royalties: HM PRO, Speakers Bureau; Daiichi: Consultancy; Sobi: Honoraria; Amgen: Honoraria, Speakers Bureau; Janssen: Consultancy; Apellis: Consultancy, Honoraria; BMS: Consultancy, Honoraria, Speakers Bureau. Rambaldi:Celgene-BMS: Honoraria; Roche: Honoraria; Incyte: Honoraria; Novartis: Honoraria; Kite-Gilead: Honoraria; Jazz: Honoraria; ABBVIE: Honoraria; Astellas: Honoraria; Pfizer: Honoraria; Amgen: Honoraria; Omeros: Honoraria; Janssen: Honoraria. Passamonti:BMS: Research Funding; Novartis, Celgene, BMS, Abbvie, Janssen, Roche, AOP Orphan, Karyopharma, KYOWA KIRIN, Mei: Consultancy, Honoraria, Speakers Bureau. Santoro:Gilead: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Pfizer: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Novartis: Speakers Bureau; Sandoz: Speakers Bureau; Bayer: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Merck Sharp & Dohme: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Sanofi: Consultancy; Eli-Lilly: Speakers Bureau; AstraZeneca: Speakers Bureau; Abb-vie: Speakers Bureau; Roche: Speakers Bureau; Bristol Myers Squibb: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Eisai: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Servier: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Celgene: Speakers Bureau; Amgen: Speakers Bureau; Takeda: Speakers Bureau; Incyte: Consultancy. Germing:Celgene: Consultancy, Honoraria, Research Funding; Novartis: Consultancy, Honoraria. Kordasti:Novartis: Membership on an entity's Board of Directors or advisory committees, Research Funding; Alexion: Speakers Bureau; BMS: Research Funding; MorphoSys: Research Funding; Beckman Coulter: Speakers Bureau. Santini:Takeda: Membership on an entity's Board of Directors or advisory committees; Novartis: Honoraria, Membership on an entity's Board of Directors or advisory committees; Menarini: Consultancy, Membership on an entity's Board of Directors or advisory committees; Gilead: Membership on an entity's Board of Directors or advisory committees; Geron: Membership on an entity's Board of Directors or advisory committees; BMS: Honoraria, Membership on an entity's Board of Directors or advisory committees; Otsuka: Membership on an entity's Board of Directors or advisory committees; Servier: Membership on an entity's Board of Directors or advisory committees; Syros: Membership on an entity's Board of Directors or advisory committees. Diez-Campelo:BluePrint: Membership on an entity's Board of Directors or advisory committees; Bristol Myers Squibb: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Novartis: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Takeda: Honoraria, Membership on an entity's Board of Directors or advisory committees. Sanz:takeda: Honoraria; Janssen Pharmaceuticals, Inc.: Other: Teaching and Speaking; La Hoffman Roche Ltd.: Other: Advisor or review panel participant; Novartis Oncology: Consultancy; Celgene Corporation: Consultancy; Abbvie Pharmaceuticals: Other: Advisor or review panel participant; Helsinn: Honoraria, Other: Advisor or review panel participant; Takeda Pharmaceuticals Ltd: Other: Advisor or review panel participant. Kern:MLL Munich Leukemia Laboratory: Current Employment, Other: Ownership. Platzbecker:Geron: Honoraria; Takeda: Honoraria; Janssen: Honoraria; Silence Therapeutics: Honoraria; Jazz: Honoraria; Abbvie: Honoraria; BMS/Celgene: Honoraria; Novartis: Honoraria. Fenaux:Janssen: Consultancy, Honoraria, Research Funding; Takeda: Honoraria, Research Funding; BMS: Consultancy, Honoraria, Research Funding; Jazz: Consultancy, Honoraria, Research Funding; Novartis: Consultancy, Honoraria, Research Funding; Abbvie: Consultancy, Honoraria, Research Funding; Celgene/BMS: Honoraria, Research Funding; Syros Pharmaceuticals: Honoraria. Haferlach:Munich Leukemia Laboratory: Current Employment, Other: Part ownership.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal